aPer the investigator’s slit lamp assessment and the following categories: lens clear, lens opacity present–not significant, and lens opacity present–visually significant.
bBased on visual field changes.
AE = treatment-emergent adverse event.
DISCOVER RESULTS FROM PIVOTAL TRIAL AND GPS:
GPS OPEN-LABEL STUDY ON XEN® GEL STENT VS TRABECULECTOMY
Results of a prospective, multicenter, single-arm, open-label, US clinical trial to evaluate the safety and effectiveness of the XEN® Gel Stent in refractory glaucoma subjects (N = 65) where previous filtering or cilioablative procedures failed, or IOP was unresponsive to maximum tolerated medication. Medication washout was not performed; all IOP-lowering medications were discontinued on the day of surgery.1
Study eyes undergoing glaucoma-related secondary surgical intervention (SSI) and/or removal of the XEN® Gel Stent prior to the 12-month evaluation were considered to be nonresponders. Seven subjects in the study underwent needling procedures with mitomycin C; 4 of these subjects were considered responders.1
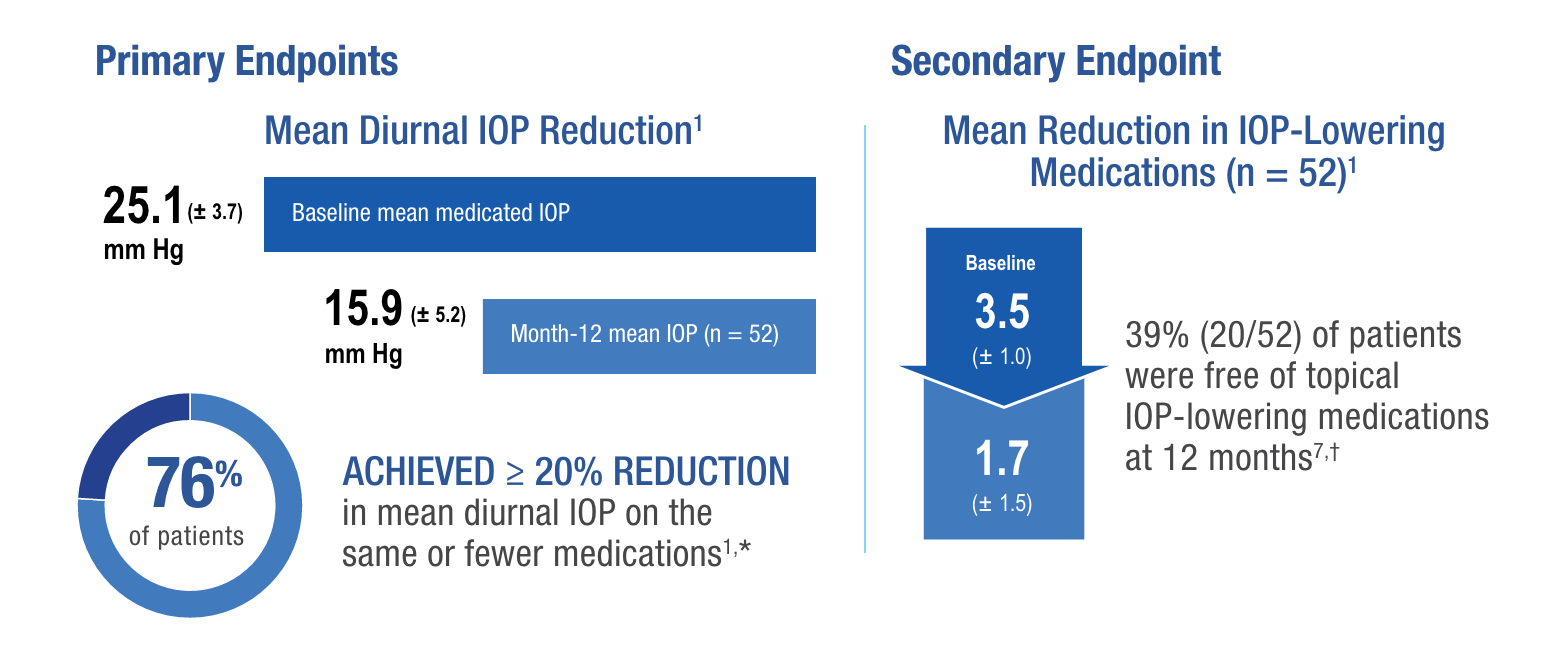
*76.3% (95% CI = 65.8%, 86.8%); using observed data and failures for subjects with glaucoma-related SSI and multiple imputations for missing data (N = 65).
†Patients who completed the 12-month visit (n = 52).
The most common postoperative AEs included BCVA loss of ≥ 2 lines (≤ 30 days 15.4%; > 30 days 10.8%; 12 months 6.2%), hypotony IOP < 6 mm Hg at any time (24.6%), IOP increase ≥ 10 mm Hg from baseline (21.5%), and needling procedure (32.3%).1

‡No clinically significant consequences were associated with hypotony, such as choroidal effusions, suprachoroidal hemorrhage, or hypotony maculopathy. IOP < 6 mm Hg was defined as an adverse event, regardless of whether there were any associated complications or sequelae related to the low pressure. Thirteen cases occurred at the 1-day visit; there were no cases of persistent hypotony, and no surgical intervention was required for any case of hypotony.1
*The XEN® Directions for Use (DFU) discusses sponge administration of MMC, an antifibrotic therapy, to the surgical site.1 Administration of MMC/antifibrotic therapy by injection is not approved in the United States.
GPS = Gold Standard Pathway Study.
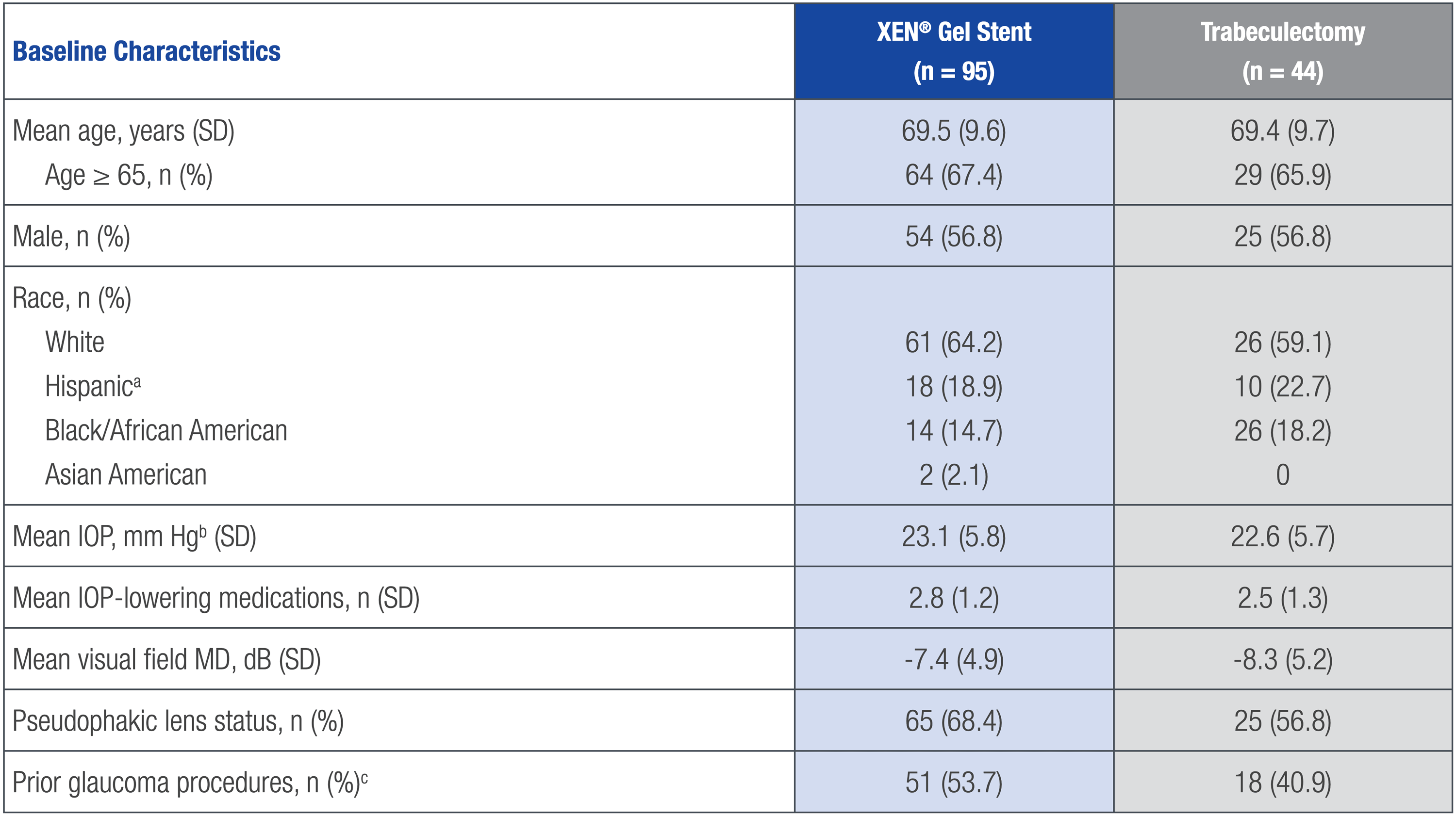
aHispanic patients have been previously presented as American Indian/Alaska Native (following the Clinical Data Interchange Standards Consortium control terminology tabulation model).
bStudy eye.
cIncluded laser procedures and surgeries (surgeries included cycloablation, minimally invasive surgery, and trabeculectomy).
A composite endpoint of patients (%) achieving the following at the 12-month visit:
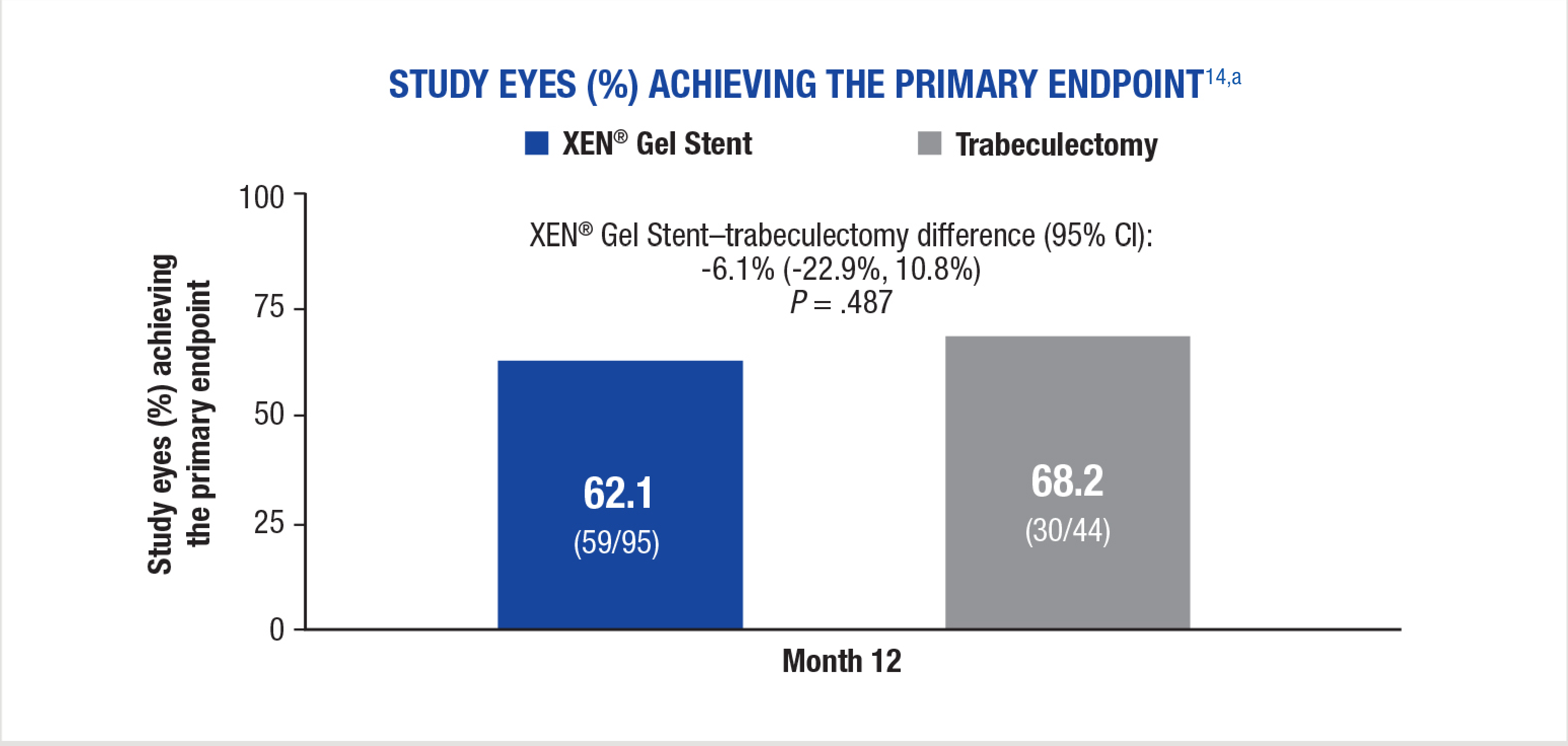
aPatients with missing data were considered failures in this analysis.15
All secondary endpoints were prespecified, nonranked endpoints, and were not controlled for multiplicity. Therefore, these secondary endpoints cannot be regarded as statistically significant.
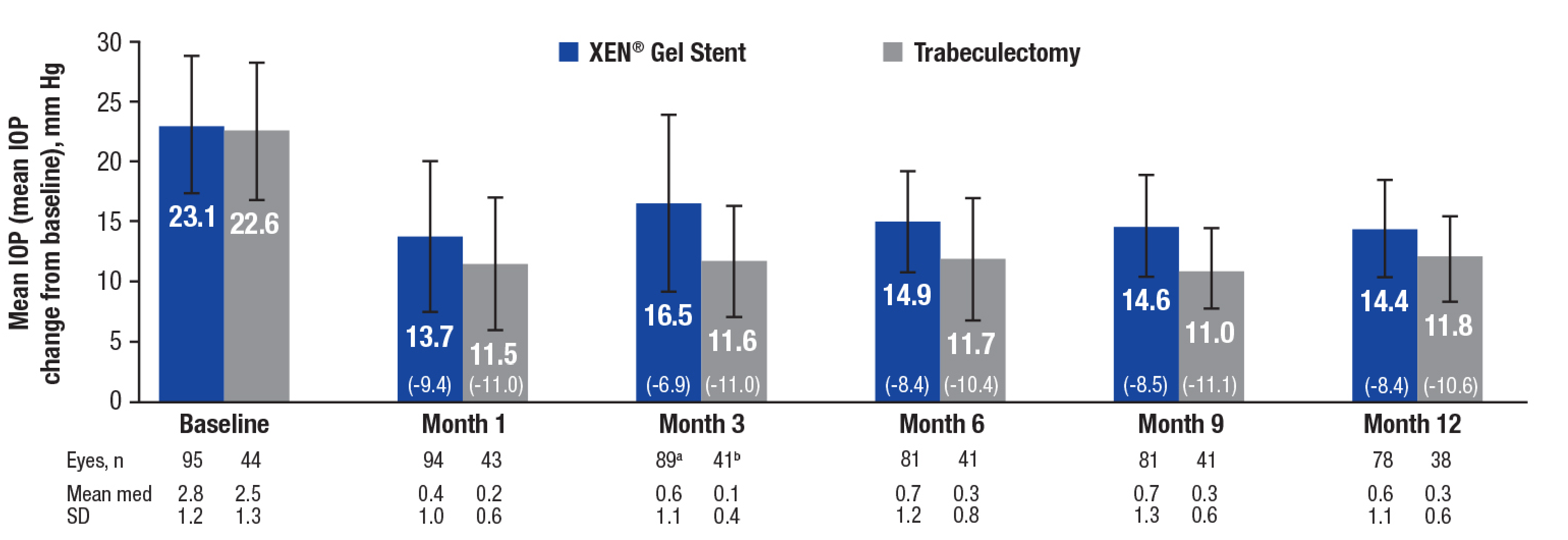
an = 91 (month 3), 82 (month 6), and 87 (month 12). bn = 42 (month 3) and 41 (month 12). Both footnotes refer to the medication count analysis.
med = medication.
All secondary endpoints were prespecified, nonranked endpoints, and were not controlled for multiplicity. Therefore, these secondary endpoints cannot be regarded as statistically significant.
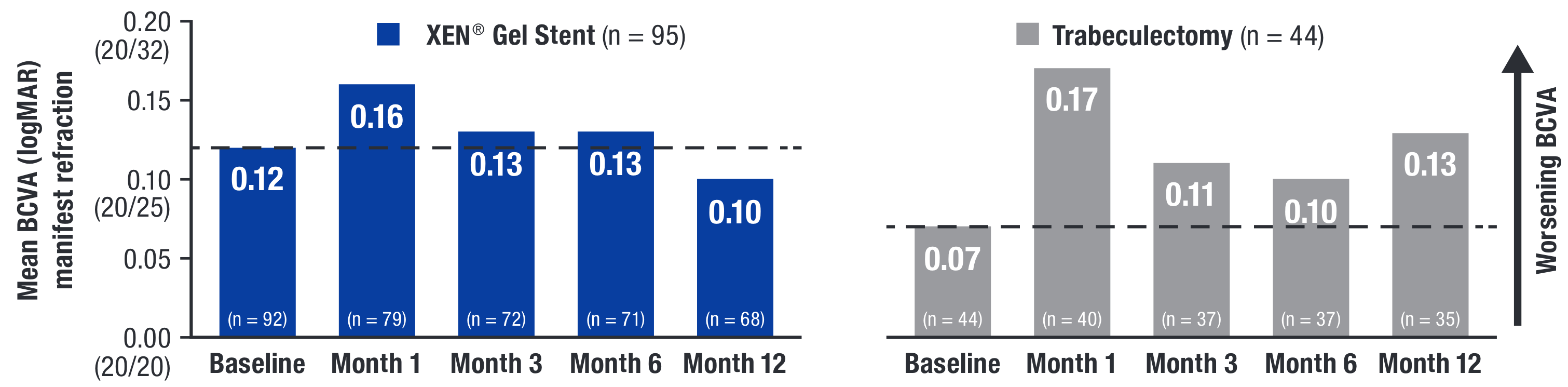
Y-axis values in parentheses correspond to equivalent ETDRS Snellen fraction conversion from logMAR units.16
ETDRS = Early Treatment Diabetic Retinopathy Study; logMAR = logarithm of the minimum angle of resolution.
PROs UTILIZING THE VALIDATED SYMPTOM AND HEALTH PROBLEM CHECKLIST (SHPC-18)14
All secondary endpoints were prespecified, nonranked endpoints, and were not controlled for multiplicity. Therefore, these secondary endpoints cannot be regarded as statistically significant.
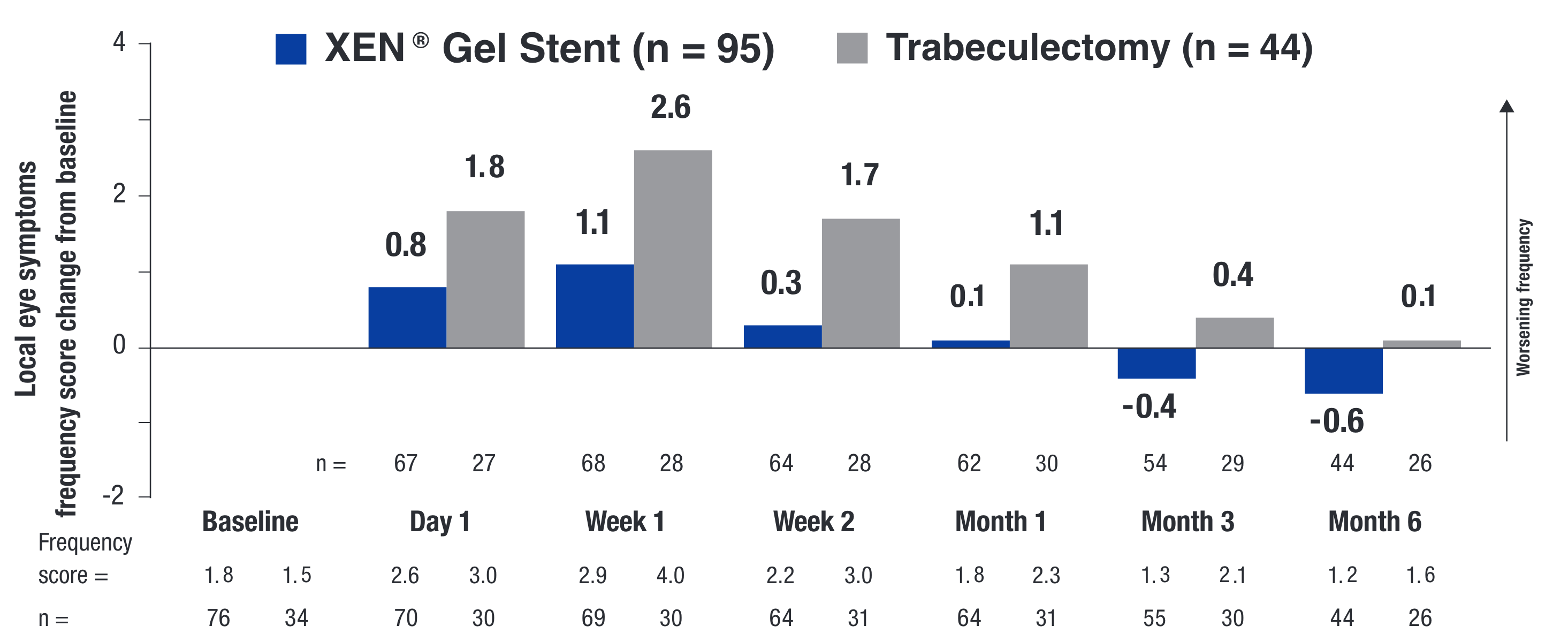
All secondary endpoints were prespecified, nonranked endpoints, and were not controlled for multiplicity. Therefore, these secondary endpoints cannot be regarded as statistically significant.
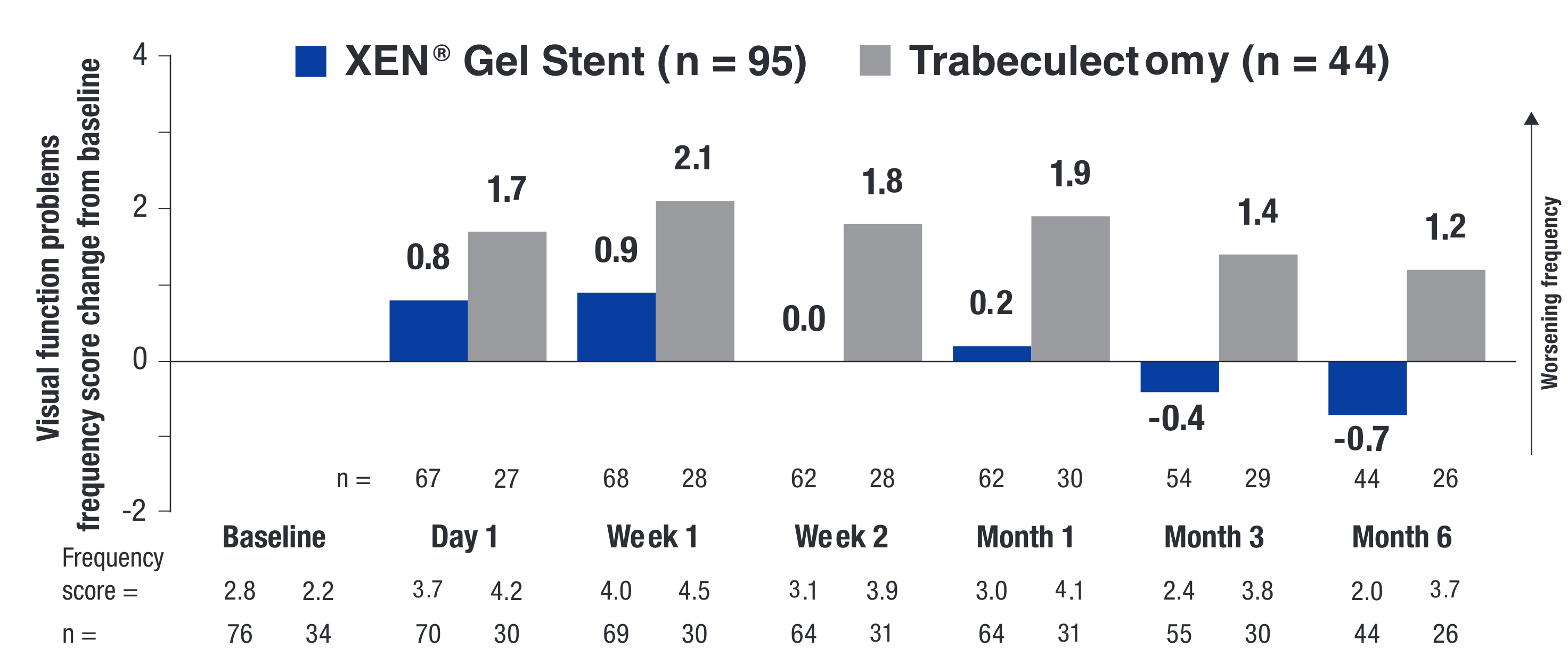
The change from baseline or between-treatment differences (∆; XEN® Gel Stent minus trabeculectomy) were calculated using least squares means for each treatment arm.
PRO = patient reported outcome.
POSTOPERATIVE INTERVENTIONS IN OFFICE14

aTwo patients received both MMC and 5-fluorouracil during needling.
bIncluded air injection in the anterior chamber (n = 2), XEN® Gel Stent removal (n = 2), laser iridotomy (n = 2), digital ocular compression (n = 1), goniosynechialysis (n = 1), laser iridoplasty (n = 1), and paracentesis (n = 1) in the XEN® Gel Stent arm, and digital ocular compression (n = 2) and suture removal (n = 3) in the trabeculectomy arm.
NA = not applicable.
OPERATIVE AND POSTOPERATIVE AEs14
–The most common postoperative ocular AEs in both treatment arms were visual acuity reduced by ≥ 2 lines, hypotony (IOP < 6 mm Hg), and IOP increased by ≥ 10 mm Hg from baseline
–The most common surgical complication was anterior chamber bleeding, reported in 1.1% (1/95) of XEN® Gel Stent patients and 4.5% (2/44) of trabeculectomy patients
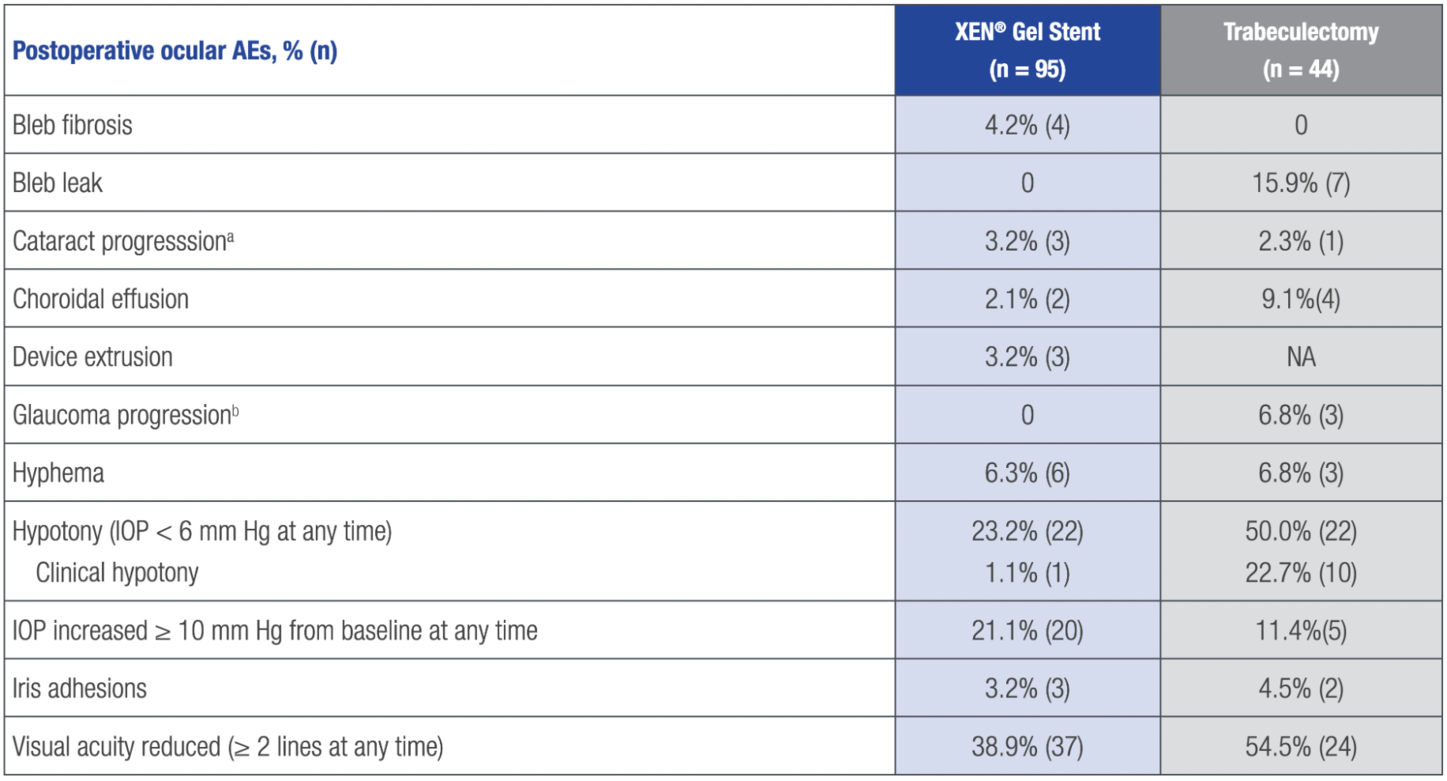
aPer the investigator’s slit lamp assessment and the following categories: lens clear, lens opacity present–not significant, and lens opacity present–visually significant.
bBased on visual field changes.
AE = treatment-emergent adverse event.
The XEN® Glaucoma Treatment System (XEN® 45 Gel Stent preloaded into a XEN® Injector) is indicated for the management of refractory glaucomas, including cases where previous surgical treatment has failed, cases of primary open-angle glaucoma, and pseudoexfoliative or pigmentary glaucoma with open angles that are unresponsive to maximum tolerated medical therapy.
XEN® Gel Stent is contraindicated in angle-closure glaucoma where angle has not been surgically opened, previous glaucoma shunt/valve or conjunctival scarring/pathologies in the target quadrant, active inflammation, active iris neovascularization, anterior chamber intraocular lens, intraocular silicone oil, and vitreous in the anterior chamber.
XEN® Gel Stent complications may include choroidal effusion, hyphema, hypotony, implant migration, implant exposure, wound leak, need for secondary surgical intervention, and intraocular surgery complications. Safety and effectiveness in neovascular, congenital, and infantile glaucoma has not been established. Avoid digital pressure following implantation of the XEN® Gel Stent to avoid the potential for implant damage.
Examine the XEN® Gel Stent and XEN® Injector in the operating room prior to use. Monitor intraocular pressure (IOP) postoperatively and if not adequately maintained, manage appropriately. Stop the procedure immediately if increased resistance is observed during implantation and use a new XEN® system. Safety and effectiveness of more than a single implanted XEN® Gel Stent has not been studied.
The most common postoperative adverse events included best-corrected visual acuity loss of ≥ 2 lines (≤ 30 days 15.4%; > 30 days 10.8%; 12 months 6.2%), hypotony IOP < 6 mm Hg at any time (24.6%; no clinically significant consequences were associated, no cases of persistent hypotony, and no surgical intervention was required), IOP increase ≥ 10 mm Hg from baseline (21.5%), and needling procedure (32.3%).
Caution: Federal law restricts this device to sale by or on the order of a licensed physician. Please call 1-800-433-8871 to report an adverse event.
Please see accompanying full Directions for Use or visit https://www.rxabbvie.com/pdf/xen_dfu.pdf
US-XEN-230091